
Faculty of the Division of Rheumatology, Inflammation and Immunity at Brigham and Women’s Hospital played a prominent role at the American College of Rheumatology’s annual Convergence conference, which ran virtually from November 3 through November 10, 2021.
Ellen M. Gravallese, MD, chief of the Division of Rheumatology, Inflammation and Immunity, noted that despite the virtual format, 2021 ACR Convergence was both well-attended (more than 14,000 attendees from about 110 countries) and informative.
“It was once again an exciting conference,” she said. “Speaking on behalf of the division, we were honored to contribute so many presentations and to help make the meeting a success.”
Meeting highlights contributed by the division included:
- Michael B. Brenner, MD, delivered the Basic Science Year in Review, and Karen H. Costenbader, MD, MPH, delivered the Clinical Science Year in Review.
- Seoyoung C. Kim, MD, ScD, MSCE, received the Henry Kunkel Early Career Investigator Award, while Alisa Mueller, MD, PhD, took home the Distinguished Fellow Award
- Costenbader presented a Plenary Session abstract and served as co-host of the SLE Community Hub
- Katherine P. Liao, MD, MPH, served as co-host of the RA Community Hub
- Deepak A. Rao, MD, PhD, delivered two presentations: “Adaptive Immune Cells in the RA Synovium,” and “T Peripheral Helper Cells in Autoimmune Disease”
- Rao, Anna Helena Jonsson, MD, PhD, and Soumya Raychaudhuri, MD, PhD, took part in the “Molecular Insights from the Accelerating Medicines Partnership 2021: RA/SLE” sessions
In all, division physicians and scientists contributed 103 abstracts, including 31 oral presentations, 71 poster presentations and one plenary presentation. The following is a look at seven of these abstracts.
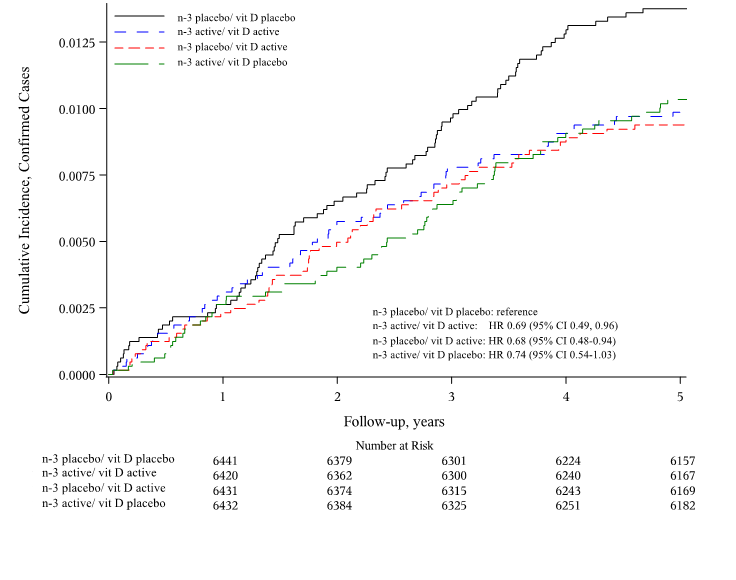
Vitamin D and Marine n-3 Fatty Acid Supplementation and Prevention of Autoimmune Disease in the VITAL Randomized Controlled Trial
 Dr. Costenbader and colleagues reported results on prevention of autoimmune diseases in the VITamin D and OmegA-3 TriaL (VITAL). In this U.S.-wide, randomized, double-blind, placebo-controlled trial, 25,871 participants were randomized to vitamin D (2000 U/d) and/or n-3 fatty acid (1000 mg/d) supplementation or placebo for five years. New doctor-diagnosed autoimmune diseases were reported and confirmed by expert physicians. Of participants, 20 percent were Black and 51 percent were women; mean age was 67.1 years. Vitamin D and/or n-3 fatty acids reduced incident autoimmune diseases by 25 percent to 30 percent.
Dr. Costenbader and colleagues reported results on prevention of autoimmune diseases in the VITamin D and OmegA-3 TriaL (VITAL). In this U.S.-wide, randomized, double-blind, placebo-controlled trial, 25,871 participants were randomized to vitamin D (2000 U/d) and/or n-3 fatty acid (1000 mg/d) supplementation or placebo for five years. New doctor-diagnosed autoimmune diseases were reported and confirmed by expert physicians. Of participants, 20 percent were Black and 51 percent were women; mean age was 67.1 years. Vitamin D and/or n-3 fatty acids reduced incident autoimmune diseases by 25 percent to 30 percent.
These results have high clinical importance, as the agents are well-tolerated and non-toxic, and no other therapies exist to prevent autoimmune diseases.
Longitudinal CyTOF Immunophenotyping Reveals Distinct Patterns of T cell-B Cell Dysregulation in SLE
Takanori Sasaki, PhD
 Early recognition of systemic lupus erythematosus (SLE) remains a major challenge for rheumatologists. Drs. Takanori Sasaki, Costenbader and Rao generated detailed immunophenotyping analyses of immune cells from patients with newly diagnosed SLE. They identified several activated T cell and B cell subsets — including T peripheral helper cells, T follicular helper cells and age-associated B cells — that are increased in patients with early lupus. They also tracked cell populations over the first year of disease to identify patterns of change over time. These studies have the potential to identify biomarkers to detect and track immune activation in patients with early SLE.
Early recognition of systemic lupus erythematosus (SLE) remains a major challenge for rheumatologists. Drs. Takanori Sasaki, Costenbader and Rao generated detailed immunophenotyping analyses of immune cells from patients with newly diagnosed SLE. They identified several activated T cell and B cell subsets — including T peripheral helper cells, T follicular helper cells and age-associated B cells — that are increased in patients with early lupus. They also tracked cell populations over the first year of disease to identify patterns of change over time. These studies have the potential to identify biomarkers to detect and track immune activation in patients with early SLE.
A Phenome-Wide Association Study of Genes Associated with COVID-19 Severity Reveals Shared Genetics with Rheumatic Conditions
 International efforts have identified several genetic variants associated with severe COVID-19. Determining whether these genes are also shared by known human conditions can further inform our understanding of COVID-19.
International efforts have identified several genetic variants associated with severe COVID-19. Determining whether these genes are also shared by known human conditions can further inform our understanding of COVID-19.
In a screen of ~1,800 phenotypes with data from the nation’s largest biobank, the Veteran Affairs’ Million Veteran Program, Dr. Liao and colleagues identified several rheumatic conditions (e.g., psoriasis and systemic lupus erythematosus) that share genes with severe COVID-19 susceptibility. Moreover, these variants were associated with a reduced risk of the rheumatic conditions. This divergent association for the same gene places a spotlight on the shared pathways of host defense and autoimmunity — and on why specific immunomodulating therapies have been effective against COVID-19.
Cellular Origin and Functions of Osteoclasts in Inflammatory Arthritis
Christian E. Jacome-Galarza, PhD
 Osteoclasts (OCs) cause bone erosion and joint destruction in inflammatory arthritis (IA), leading to physical disability. The precise cellular origins of OCs are poorly understood.
Osteoclasts (OCs) cause bone erosion and joint destruction in inflammatory arthritis (IA), leading to physical disability. The precise cellular origins of OCs are poorly understood.
Using a variety of cell lineage tracing systems, Dr. Jacome-Galarza and colleagues found new evidence that OCs can differentiate from distinct cellular sources in the inflamed synovium. These systems will facilitate the identification of unique differences among OCs of distinct origins. These differences, in turn, will allow for differential targeting of OCs via unique cell surface markers and for the development of new targets to inhibit OC formation and prevent bone erosions in IA.
Predictors of Rheumatic Immune-related Adverse Events After Immune Checkpoint Inhibitor Use for Cancer
 Immune-related adverse events (irAEs) are common toxicities resulting from the blockade of immune checkpoint inhibitors (ICIs) for cancer treatment, and some resemble primary rheumatic diseases. Under the supervision of Dr. Sparks, Brigham researchers identified all cancer patients initiating ICIs at Mass General Brigham and Dana-Farber Cancer Institute (n=8,028). They systematically identified all rheumatic irAE cases using stringent criteria by record review (n=226) and performed a case-control study to identify predictors of rheumatic irAEs at the time of ICI initiation.
Immune-related adverse events (irAEs) are common toxicities resulting from the blockade of immune checkpoint inhibitors (ICIs) for cancer treatment, and some resemble primary rheumatic diseases. Under the supervision of Dr. Sparks, Brigham researchers identified all cancer patients initiating ICIs at Mass General Brigham and Dana-Farber Cancer Institute (n=8,028). They systematically identified all rheumatic irAE cases using stringent criteria by record review (n=226) and performed a case-control study to identify predictors of rheumatic irAEs at the time of ICI initiation.
Dr. Sparks and colleagues found that melanoma had four-fold higher odds and genitourinary cancer had two-fold higher odds for rheumatic irAEs than lung cancer. Additionally, combination ICI use, pre-existing autoimmune disease and recent glucocorticoid use were each associated with higher odds of rheumatic irAEs. These results may help clinicians identify patients at risk of rheumatic irAEs and will lead to future epidemiologic and biomarker studies.
Risk of Cardiovascular Outcomes in Patients Treated with Tofacitinib: First Results from the Safety of TofAcitinib in Routine Care Patients with Rheumatoid Arthritis (STAR-RA) Study
 The post-marketing ORAL Surveillance safety trial has raised concerns regarding the cardiovascular safety of tofacitinib among patients with rheumatoid arthritis (RA). Using a multi-database population, Dr. Kim and colleagues evaluated the cardiovascular safety of tofacitinib compared to TNF inhibitors in a “real-world evidence” (RWE) cohort consisting of adult patients with RA from routine care and an “RCT-duplicate” cohort including RA patients at high cardiovascular risk similar to the ORAL Surveillance trial.
The post-marketing ORAL Surveillance safety trial has raised concerns regarding the cardiovascular safety of tofacitinib among patients with rheumatoid arthritis (RA). Using a multi-database population, Dr. Kim and colleagues evaluated the cardiovascular safety of tofacitinib compared to TNF inhibitors in a “real-world evidence” (RWE) cohort consisting of adult patients with RA from routine care and an “RCT-duplicate” cohort including RA patients at high cardiovascular risk similar to the ORAL Surveillance trial.
The investigators found no increased cardiovascular risk associated with tofacitinib in the RWE cohort (HR 1.01, 95 percent CI, 0.83-1.23). However, consistent with the result from ORAL Surveillance, tofacitinib was associated with a numerically increased cardiovascular risk in the RCT-duplicate cohort (HR 1.24, 95 percent CI, 0.90-1.69).
Risk of Major Adverse Cardiovascular Events in a Large Cohort of Patients with Acute Calcium Pyrophosphate Crystal Arthritis
 Many types of inflammatory arthritis are associated with increased risk for cardiovascular disease. Dr. Tedeschi and colleagues studied the risk of cardiovascular events in patients with pseudogout, an acute inflammatory crystalline arthritis.
Many types of inflammatory arthritis are associated with increased risk for cardiovascular disease. Dr. Tedeschi and colleagues studied the risk of cardiovascular events in patients with pseudogout, an acute inflammatory crystalline arthritis.
In a cohort of 1,200 Mass General Brigham patients with pseudogout, the risk of myocardial infarction, coronary revascularization, acute coronary syndrome or stroke was significantly higher than in comparators after adjusting for traditional cardiovascular risk factors (HR 1.92 in years 0-2 after diagnosis; HR 2.18 in years 2-10). Results were similar when the analysis was restricted to non-hospitalized patients and among patients with linked Medicare claims data.