Lung diseases like chronic obstructive pulmonary disease (COPD) can be remarkably diverse in their pathologies and the ways in which patients respond to treatment. To better understand diseases such as COPD, pulmonary vascular disease and interstitial lung disease, researchers at Brigham and Women’s Hospital created the Applied Chest Imaging Laboratory (ACIL). The lab leverages the power of imaging and hypothesis-driven modeling to create algorithms that empower clinical and genetic research.
The ACIL’s co-creators and co-directors are George Richard Washko Jr., MD, of the Brigham’s Pulmonary and Critical Care Medicine Division and Raúl San José Estépar, PhD, associate professor of radiology at Harvard Medical School.
They started their lab about 15 years ago as they came out of postgraduate training and saw an opportunity to leverage medical image analysis in clinical investigation. With the strong support of their respective mentors in pulmonology and radiology, they began performing secondary analyses on clinical- and research-acquired CT scans of the chest. That work led to conferences, presentations, publications and ultimately collaborations at the local, national and international levels. Today, the lab has about 18 members, half of whom are based in radiology and half of whom are based in medicine.
“This cross-disciplinary effort has been a great strength for our group, with clinicians working with computer scientists to address clinical questions and computer scientists working with clinicians to develop and optimize software tools to provide new insights into disease,” Dr. Washko said. “We can solve problems through bi-directional processes not seen in other imaging labs.”
The Emerging Power of Imaging Data in Pulmonology
While imaging has become a common therapeutic tool for pulmonary device interventions, such as lung volume reduction, its role in understanding the functional implications of lung structure and changes in disease pathology is just emerging. According to Dr. Washko, imaging’s ability to visualize injury patterns in the lungs allows clinicians to categorize patients into subtypes and match those subtypes with personalized treatment options that will result in the best outcomes.
“Based on the visualized structure of the chest, we can identify those patients who are more resilient to adverse events and those who are more susceptible,” he said. “We can also use imaging to objectively determine response to treatment long before it is clinically evident.”
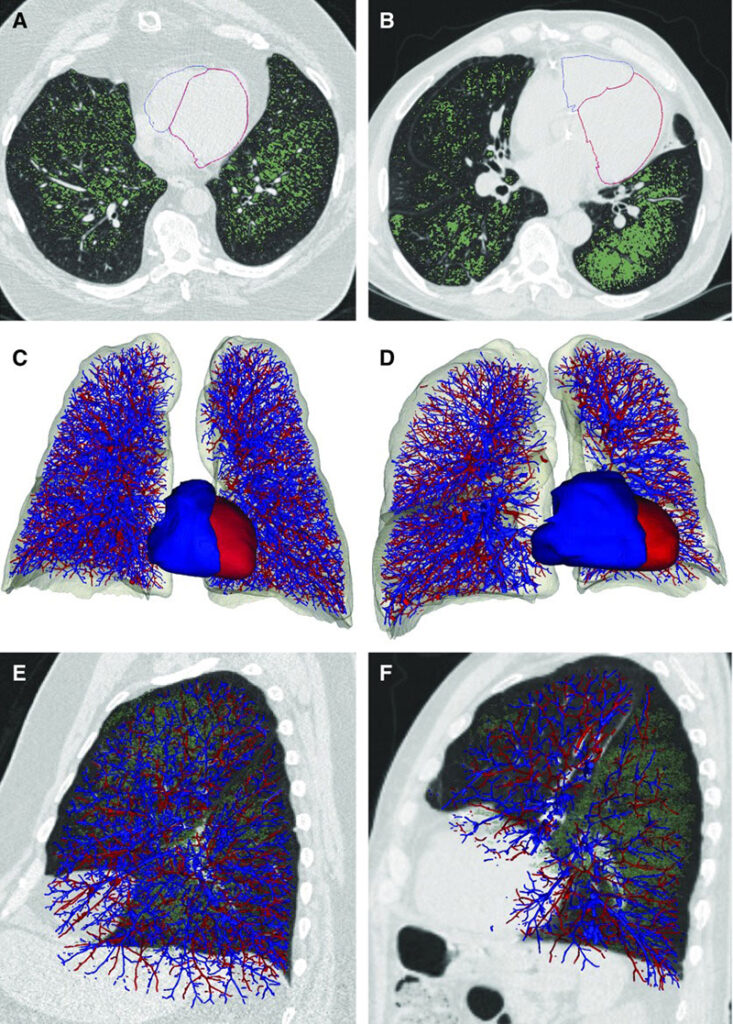
Dr. Estépar and his colleagues take this information further by crunching the vast amount of imaging data they have collected — terabytes of data from up to 50,000 scans — and creating complex algorithms that can be scaled up for future studies. They have created an open source toolkit called the Chest Imaging Platform, which is available to the larger pulmonary community and provides a friendly clinical entry point to the group’s methods.
“Our lab is one of the few groups in the country that can process, analyze and scale such large amounts of data and then translate that data for clinical studies,” Dr. Estépar said. “It may sound trivial, but this level of precision is extremely difficult.”
As Dr. Washko explained, the inability to scale from tens to tens of thousands had traditionally left imaging without a seat at the table in large-scale clinical trials. With the ACIL’s toolkit, image analytic techniques can now match biologic-based efforts.
“We can use imaging to power genome-wide association studies of different disease subtypes,” he said.
A Multidisciplinary Consortium of Investigators
As the ACIL has grown, team members have focused on different disease states and different organs in the chest. Some members focus on the pulmonary and vasculature and how CT can be used to detect vascular injury and measure disease progression and response to therapeutic intervention. Since these diseases and their corresponding anatomic structures provide the foundation for career development, more junior investigators ultimately “own” aspects of the image analytics.
“Our group functions as a consortium of investigators, each bringing ideas and grants to the group,” Dr. Washko said. “We also serve as a core for multiple national and international studies.”
One of the lab’s most recent research initiatives is collecting and examining pre-operative CT scans that are used to plan lung cancer surgery. Another initiative is focused on extracting imaging features related to co-morbidities to determine if imaging can augment data science efforts focused on aggregating and modeling information in the electronic health record.
The ACIL’s success has been based, in part, on its collaboration between departments, according to Dr. Washko, who said that respiratory medicine is not just about pulmonologists delivering clinical care.
Dr. Estépar echoed those sentiments and noted that the field of medicine can sometimes suffer from falling into silos — an issue the Brigham has avoided by design.
“Cross collaboration needs to be fostered,” he said. “At the Brigham, we are fortunate to have the vision and the mentors to break down walls between departments and drive innovation and mission success from different perspectives.”
*Image Citation
Authors: George R. Washko, Pietro Nardelli, Samuel Y. Ash, Gonzalo Vegas Sanchez-Ferrero, Farbod N. Rahaghi, Carolyn E. Come, Mark T. Dransfield, Ravi Kalhan, MeiLan K. Han,, Surya P. Bhatt, J. Michael Wells, Carrie Pistenmaa Aaron, Alejandro A. Diaz, James C. Ross, Michael J. Cuttica, Wassim W. Labaki, Gabriela Querejeta Roca, Amil M. Shah, Kendra Young, Gregory L. Kinney, John E. Hokanson, Alvar Agustí, Raúl San José Estépar
Article Title: Arterial Vascular Pruning, Right Ventricular Size, and Clinical Outcomes in Chronic Obstructive Pulmonary Disease. A Longitudinal Observational Study
Journal Title: American Journal of Respiratory Critical Care Medicine
Date/Volume/Page Range: 454-461 Figure 1. Volume Number: August 15, 2019 200(4) Published online 2019 Aug 15. doi: 10.1164/rccm.201811-2063OC