Ronald L. Neppl, PhD, a molecular biologist in the Department of Orthopedic Surgery at Brigham and Women’s Hospital, studies the molecular mechanisms that regulate the balance between anabolic and catabolic processes, and how imbalances in these physiological processes may lead to muscle atrophy.
“Gaining a better understanding of anabolic and catabolic processes is the first step toward developing interventions that could mitigate muscle loss and improve health span in humans,” says Dr. Neppl, who is also a principal investigator in the Musculoskeletal Research Center at the Brigham Research Institute.
Molecular Regulation of Skeletal Muscle Homeostasis in Health and Disease
In the context of muscle atrophy and hypertrophy, many factors that regulate protein synthesis (e.g., Akt1, mTor, p70S6K) and protein degradation (e.g., Atrogin-1, MuRF1, MUSA) have been well-studied, but researchers know less about how non-coding RNAs (ncRNAs) regulate skeletal muscle homeostasis and repair.
A type of ncRNA, long non-coding RNAs (lncRNAs) are genes that regulate the expression of protein-coding genes. A wide body of research has shown that lncRNAs play key roles in maintaining the homeostasis of skeletal muscles and may be involved in the progression of a disease state resulting in muscle atrophy.
“The study of lncRNAs represents an exciting area of research in the field of skeletal muscle growth and repair, as these genes have been identified as critical regulators of cellular proliferation, differentiation and senescence,” says Dr. Neppl.
lncRNA-Mediated Regulation of Skeletal Muscle Homeostasis and Repair
A major focus of the Neppl lab is understanding how ncRNAs, particularly lncRNAs, control skeletal muscle development, myogenic repair and hypertrophic growth, and how perturbations in these processes may influence health and disease.
To elucidate the roles that ncRNAs—including lncRNAs—play in maintaining lean muscle mass, Dr. Neppl and his colleagues use biochemical and molecular biology techniques, in vivo and in vitro model systems, as well as next-generation RNA sequencing.
In experiments to better understand how lncRNAs regulate muscle function, the Neppl lab recently knocked down lncRNAs in a cell culture model and observed impaired myogenesis—the process by which skeletal muscle repairs itself.
“We think that lncRNAs play key roles in regulating skeletal muscle’s endogenous stem cell population, which repairs damaged muscle after injury. We recently identified a novel lncRNA that’s conserved between mice and humans and are conducting studies in a knockout mouse model,” says Dr. Neppl.
Alternative Splicing Dysregulation May Drive Muscle Atrophy
Another area of focus in the Neppl lab is understanding how alternative splicing regulates the loss of muscle mass. Alternative splicing is a process that enables a single gene to be expressed as different protein variants, or isoforms, that may have different cellular functions or properties.
“Our lab is interested in how alterations in alternative splicing modulate the cellular processes essential for normal muscle functionality. In our research, we discovered that alternative splicing can become dysregulated with age and may be contributing to the loss of muscle mass. If genes are not spliced correctly or if the fidelity of normal splicing is impaired, the end result can be the loss of strength and muscle mass,” says Dr. Neppl.

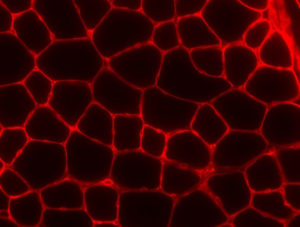
To investigate this association further, Dr. Neppl and his colleagues knocked out a gene important for regulating alternative splicing in the skeletal muscle of mice. With this gene missing, many physiological functions were impaired due to defects in normal splicing. For example, these knockout mice displayed a blunted response to hypertrophic stimuli and signs of premature aging (Image 2).
“At three months of age, these mice had the muscle mass of a two-year-old mouse. They were 20 percent smaller, weaker and couldn’t run as fast or as far. They also developed premature kyphosis with associated loss in bone density,” says Dr. Neppl.
Resistance Training: Only Intervention Proven to Maintain Lean Muscle Mass
The aging process is a multifactorial, complex process and muscle atrophy is linked with a myriad of cellular and molecular mechanisms. But are there any proven ways of slowing the age-associated loss of muscle mass?
“Resistance training is the only intervention that’s clinically proven to slow the loss of muscle mass with age. According to research, resistance training decreases the risk of chronic disease, premature morbidity and even mortality,” says Dr. Neppl.
The mechanisms by which resistance training maintains lean muscle mass involves the increase in blood flow and the flow of nutrients to skeletal muscles. Resistance training also stimulates an increase in the growth factor, IGF-I, which declines with age.
Moving Toward Biomarkers that Identify the Risk of Severe Muscle Loss
Going forward, the Neppl lab aims to continue studying lncRNAs and alternative splicing regulation in order to better understand the processes that promote the age-associated decline of skeletal muscle, particularly in conditions like sarcopenia.
In ongoing basic research, Dr. Neppl and his colleagues have identified signal transduction cascades, cellular stress responses and autocrine and paracrine factors that are sensitive to changes in alternative splicing and lncRNA expression.
“Much work still needs to be done, but we envision that lncRNAs or alternatively spliced transcripts may one day be used as biomarkers to identify individuals who are at risk of severe muscle loss,” says Dr. Neppl.